Marine
Clyde 2020 – Sampling the sediment
October 11, 2017 by Marine Directorate Communications No Comments | Category Fisheries, Freshwater Fisheries, Marine Directorate Science, Research Vessel Surveys
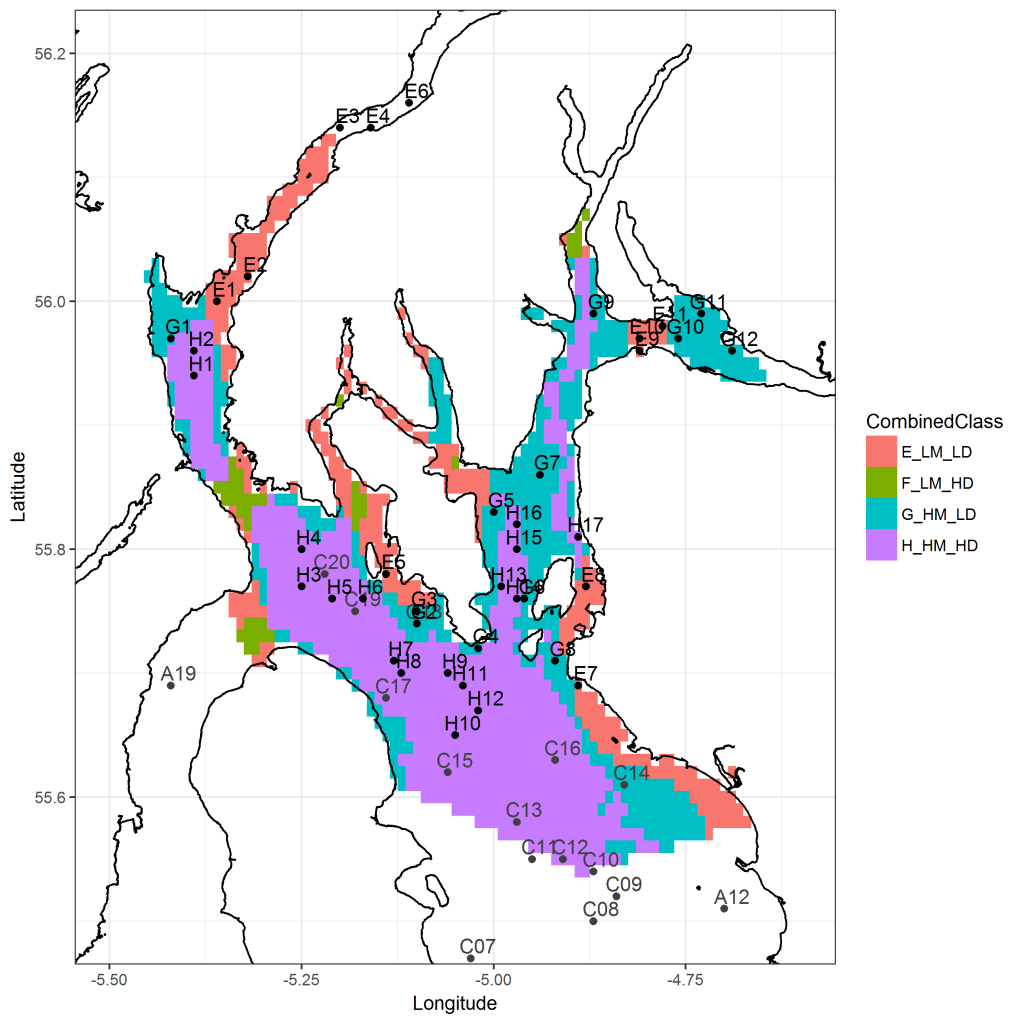
Figure 1. Map showing the proposed sampling stations (Black points) and stations sampled in April 2017 (grey points) in the Firth of Clyde superimposed over gridded stratified clusters of high and low mud content (>25%) and depth (50m).
E_LM_LD: Low Mud / Low Depth, F_LM_HD: Low Mud / High Depth, G_HM_LD: High Mud / Low Depth, H_HM_HD: High Mud / Low Depth.
Duration: 13-17 October 2017
Fishing Gear:
- Craib corer
- Day Grab – 0.1m2
- Autosieve (0.5mm & 1mm mesh sieves)
- CTD
Objectives:
- To map spatial distribution of sediment characteristics, infauna composition, pore-water nutrients and organic matter content within the inner Firth of Clyde, Clyde sea lochs and Clyde Estuary.
- To map the relationship between mean particle grain size and permeability/porosity in the Firth of Clyde.
- Determine how relationships among sediment properties and sediment nutrient content are modified by natural disturbance, fishing activity and bioturbation potential.
Procedure:
Figure 1 shows the proposed stations from which grab and core samples of seabed sediment will be collected. Stations are stratified according to depth and sediment mud content (see Figure 2).
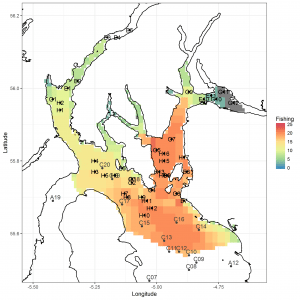
Figure 2: Maps of the Firth of Clyde superimposing proposed sampling stations (Black points) and stations sampled in April 2017 (grey points) over spatial distributions of fishing effort (number of vessels) for vessels < 15 m. ScotMap data.
Stations in the Clyde Estuary and in the vicinity of the isles of Bute and Cumbrae will be sampled Day 1 (13 October). Subsequent sampling days will then focus on sampling stations south of Bute and east of Arran, and stations west of Bute and in Loch Fyne. It is expected that ~ 5 stations will be sampled per day.
The survey anticipates equal emphasis on grab sampling and coring. The target would, therefore, be six cores, two grab samples and one CTD profile from each station.
Grab sampling
Two replicate grab samples will be collected per station. Subsamples will be taken from each grab samples and frozen for subsequent laboratory analysis of grain size distribution, % TOC/TN, sediment Chlorophyll-a & Phaeophytin-a content. The remaining sample will be sieved through a 0.5 mm mesh and the retained material preserved in 4% Saline Formaldehyde for subsequent laboratory extraction and identification of benthic invertebrates. Samples will augment data from SEPA and SNH, and provide estimates of benthic biomass for hitherto unsampled areas.
Coring
Coring may not be possible in sediments with low mud content and high tidal currents. Hence, coring will be attempted until three hours elapse or six cores are collected.
Assuming the collection of six replicate cores per station, three will analysed for permeability, porosity and grain size distribution, and three for pore-water nutrients (Ammonium, Phosphates, Silicates and Nitrates), with the extraction of four pore-water nutrient samples and 3 measurements of porosity per core.
Sampling 20 stations would yield:
- (Core samples from 10 stations) 60 cores
- 30 overlying water nutrient samples
- 90 pore-water nutrient samples
- 30 overlying water Chl-a and Pheo-a
- 30 measurements of permeability
- 90 measurements of porosity
- 180 measurements of PSA
- (Grab samples from 20 stations) 40 grab samples
- 40 PSA samples
- 40 %TOC/TN samples
- 40 Sediment Chlorophyll-a and Phaeophytin-a content
- 40 Benthic infauna samples
Ancillary Information – Chemicals to be Carried
- 4% Formaldehyde – Seawater solution (~20L) – 40% aqueous Formaldehyde is diluted with seawater and stored above 5oC.
- Di-sodium tetraborate 10-hydrate GPR (Borax) – Added at a concentration of 1g / 500ml of formosaline. Serves to prevent dissolution of bivalve shells by neutralising acidity.
- Acetone.
Further Information
Tags: Firth of Clyde, research, sample


Leave a comment